Botulinum Toxin Type A in Orofacial Pain Management: A Clinical Review
Evidence-based clinical review of botulinum toxin type A for temporomandibular disorders, myofascial pain, and bruxism-associated orofacial pain. Covers RCT evidence, injection protocols, dosing guidelines, safety, and integration with conventional therapies.
Postgraduate Level
Equivalence to 1 CPD/CME point — we do not award CPD/CME directly
Read the article, complete a short assessment, and submit your reflection to receive your AiCE Points certificate.
Take Assessment & Get CertificateLearning Objectives
- •Understanding the evidence base for BoNT-A in TMD and orofacial myofascial pain
- •Dosing protocols for masseter, temporalis, and lateral pterygoid muscles
- •Comparing BoNT-A efficacy against splints, physiotherapy, and pharmacotherapy
- •Recognising safety considerations including bone density changes with repeated use
- •Integrating BoNT-A within a multimodal orofacial pain management algorithm
This clinical review examines the off-label use of botulinum toxin type A (BoNT-A) for orofacial pain management, focusing on temporomandibular disorders (TMD) and myofascial pain. Evidence from multiple RCTs and meta-analyses demonstrates modest short-term analgesic benefit at 1 month (approximately 1.7 points on a 0–10 VAS), though long-term superiority over placebo is unproven. The review covers injection protocols for masseter, temporalis, and lateral pterygoid muscles, with dosing typically 20–50 U per masseter and 10–30 U per temporalis bilaterally. BoNT-A is positioned as a second-line treatment for refractory cases, best integrated within multimodal management.
Introduction and Clinical Overview of Orofacial Pain
Orofacial pain encompasses pain arising from the teeth, jaws, face, and related structures, often presenting as temporomandibular disorders (TMD) or myofascial pain involving the masticatory muscles. TMD is a broad term covering musculoskeletal and joint-related pain of the jaw. It affects approximately 5–12% of the population (with some studies citing up to 30–44% prevalence) and is more common in women.
Patients typically report pain in the jaw, temples, or preauricular area, which may be accompanied by jaw dysfunction (limited opening or locking), joint sounds (clicking), and even referred symptoms like headache, ear pain or fullness, and neck pain. Orofacial pain can significantly impact quality of life and daily function.
Subtypes of Orofacial Pain (TMD)
Myofascial TMD (Muscle Pain)
Pain originates from hyperactivity or strain in the masticatory muscles (masseter, temporalis, medial/lateral pterygoids). This often presents as diffuse, aching jaw/facial pain with trigger points and can refer pain to the head, face, or neck. Bruxism (teeth grinding/clenching) is a common contributing factor that can induce chronic myofascial pain.
Arthrogenic TMD (Joint Pain)
Pain arises from the temporomandibular joint (TMJ) itself, due to internal derangements (disc displacement), arthritis, or injury. This pain is localised to the TMJ area and often associated with joint noises or locking. This review focuses on muscular orofacial pain rather than intra-articular pain.
Combined Orofacial Pain
Many patients have both muscle and joint involvement, plus potential contributions from cervical (upper neck) muscle tension. Pain in the upper neck and base of skull can be part of the TMD spectrum or a related cervicogenic headache.
Conventional Therapies
First-line management of TMD and orofacial myalgia is typically conservative and multimodal. This includes patient education and behavioural modification (jaw rest, avoiding excessive chewing, stress reduction), oral appliances (occlusal stabilisation splints or night guards) to reduce bruxism forces, pharmacotherapy (nonsteroidal anti-inflammatory drugs, acetaminophen, muscle relaxants, or low-dose tricyclic antidepressants for chronic pain), and physiotherapy (jaw exercises, massage, hot/cold therapy).
In refractory cases, more invasive options like trigger point injections with local anaesthetic or corticosteroids, dry needling, or even TMJ procedures (arthrocentesis) may be tried. However, variable response rates and the chronic nature of TMD pain have led clinicians to explore other modalities – including off-label use of botulinum toxin type A.
Rationale for Botulinum Toxin (BoNT-A) in Orofacial Pain
Botulinum toxin type A (BoNT-A, e.g. Botox®) is a neurotoxin that causes reversible chemodenervation of muscles by blocking acetylcholine release at the neuromuscular junction. In the context of TMD and myofascial pain, targeted BoNT-A injection produces a dose-dependent reduction in muscle contraction and spasm of overactive masticatory muscles. By weakening the masseter, temporalis, or lateral pterygoid muscles, BoNT-A can break the cycle of clenching-related pain and allow overused muscles to recover.
Beyond muscle relaxation, BoNT-A has been shown to have direct analgesic effects: it inhibits the release of pain mediators such as substance P, calcitonin gene-related peptide (CGRP), and glutamate from peripheral nerve endings. This neuromodulatory action can reduce neurogenic inflammation and pain signalling. There is also evidence that axonal transport of BoNT-A to the trigeminal ganglion/CNS may disrupt central pain processing, contributing to pain relief.
⚠️ Regulatory Note
BoNT-A is not formally approved for TMD or orofacial pain; its use in this arena is off-label. However, given BoNT-A's success in conditions like cervical dystonia, bruxism, and migraine, clinicians have hypothesised similar benefit in TMD.
Clinical Trial Evidence: Efficacy of BoNT-A
BoNT-A vs Placebo in TMD Myofascial Pain
Several double-blind, placebo-controlled RCTs have investigated BoNT-A injections in patients with TMD-associated myofascial pain. A 2020 systematic review and meta-analysis by Machado et al included 12 RCTs and found that BoNT-A was "slightly more effective than placebo" for pain relief at 1 month, with a mean pain score improvement of about 1.7 points on a 0–10 scale (95% CI, –2.94 to –0.54). By 3 months and 6 months post-injection, however, pain scores were no longer significantly different from placebo.
Similarly, a 2024 meta-analysis (Saini et al, PLOS ONE) pooling 4 RCTs found no significant difference in pain reduction between BoNT-A and placebo at 3 and 6 months. These reviews underscore that short-term pain reduction is observed (often within 1 month) but longer-term efficacy compared to placebo remains unproven.
Key Evidence Findings
BoNT-A vs Occlusal Splints, Physiotherapy, and Medications
- BoNT-A vs Behavioural Therapy/Splint: Al-Wayli (2016) showed BoNT-A was superior at 1, 3, and 12 months compared to oral appliance use and behavioural self-care. A 2019 trial also reported greater pain relief at 3 months than stabilisation splint with home exercises.
- BoNT-A vs Physiotherapy: Guarda-Nardini et al (2012) found manual therapy (fascial manipulation) was statistically and clinically more effective than BoNT-A at 3 months for jaw myofascial pain.
- BoNT-A vs Low-Level Laser: De Carli et al (2017) found BoNT-A produced larger improvements at 1 month than low-level laser treatments, though differences evened out by 3 months.
- BoNT-A vs NSAIDs: Consensus from reviews indicates BoNT-A provides at least comparable pain relief to oral medications, without systemic side effects (no sedation or GI irritation).
In summary, BoNT-A's pain relief tends to manifest within 1–4 weeks post-injection, peaking around 6–8 weeks, whereas therapies like splints or physiotherapy may require longer consistent use. BoNT-A could be considered a faster-acting intervention for short-term relief, while conventional therapies address long-term functional improvement.
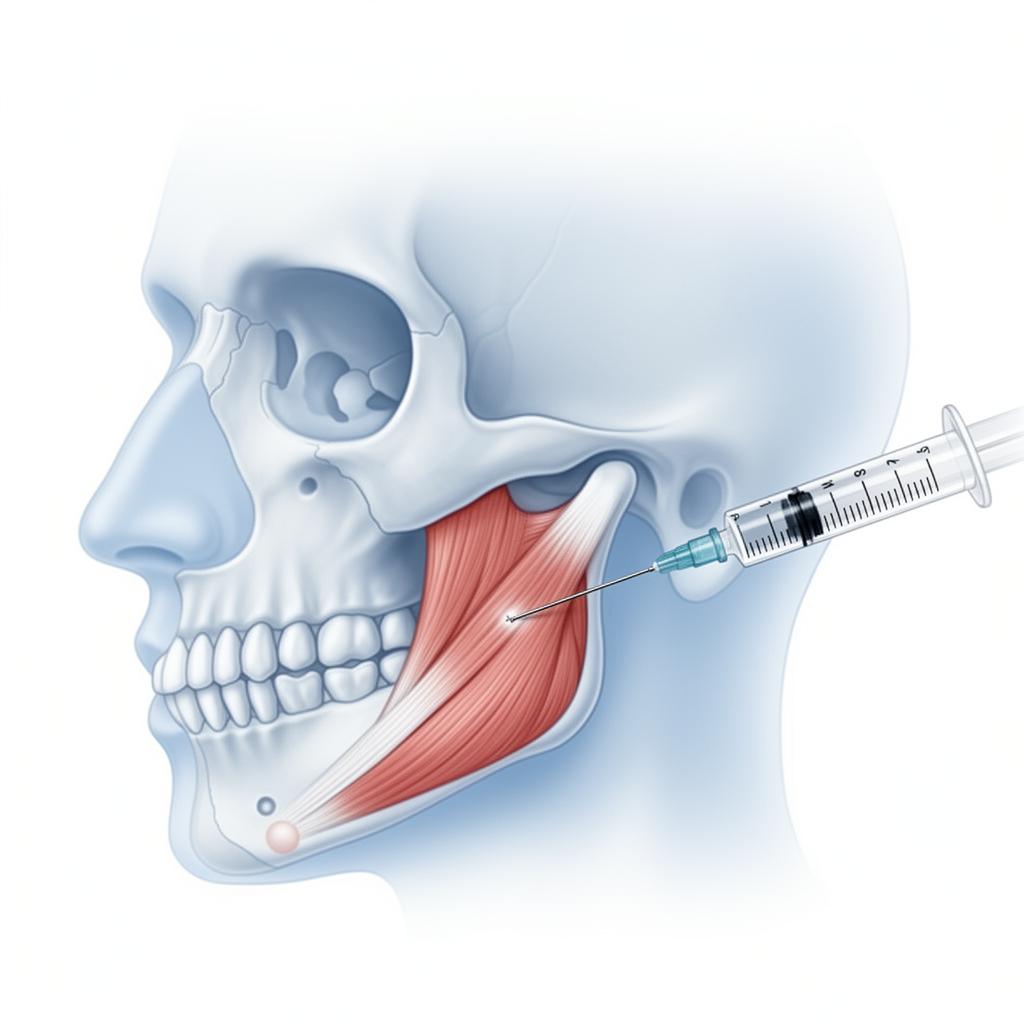
Injection Protocol: Dosage, Muscles Targeted, and Technique
| Muscle | Typical BoNT-A Dose Per Side | Injection Pattern | Repeat Interval |
|---|---|---|---|
| Masseter | ~20–30 U (female); ~30–50 U (male). High ranges 50–60 U for very hypertrophic muscles. Total 40–100 U bilaterally. | 2–4 points into mid-belly, focusing on muscle bulge during clenching. Avoid parotid gland region. | ~3 months. Clinical effect wanes by 3–4 months. Repeat no sooner than 12 weeks. |
| Temporalis | ~10–20 U (female); ~15–30 U (male). Often about half the masseter dose. Total 20–60 U bilaterally. | 2–3 points: mid-belly (above ear) and anterior portion (near hairline). Avoid superficial temporal artery. | ~3 months. Monitor for temple muscle atrophy (hollowing). |
| Lateral Pterygoid | ~5–15 U per side (starting 10 U). Up to 20–25 U for severe cases. Total 10–30 U bilaterally. | Usually 1 injection per muscle. Intra-oral or percutaneous approach via sigmoid notch. EMG or ultrasound guidance recommended. | ~3 months. Trial basis initially. Careful with cumulative dose near TMJ structures. |
Dose Individualisation
Because muscle thickness and volume vary, clinicians often start at a conservative dose and titrate in subsequent sessions based on response. Mor et al (2015) recommend ~25 U per masseter and 10 U per temporalis as a reasonable starting point for women, and higher for men (40–50 U masseter). Doses below 20 U in the masseter are often insufficient for meaningful pain relief. The FDA-mandated maximum total dose of onabotulinumtoxinA is 400 U in any 3-month interval.
Injection Technique
BoNT-A is delivered via intramuscular injections using a dilution of 1–2 mL saline per 100 U vial (5–10 U per 0.1 mL). Key points include: identifying the target muscle belly by asking the patient to clench, using a fine-gauge needle (30G), and distributing the toxin across several sites. The masseter is injected at three points across the muscle – anterior, middle, and posterior aspects – to cover the entire superficial masseter.
Injection of the lateral pterygoid requires advanced technique: this deep muscle is not accessible externally. The intra-oral approach inserts the needle behind the maxillary tuberosity region at a superior-medial angle. Because critical structures lie nearby, electromyography (EMG) or ultrasound guidance is often employed. Generally, lateral pterygoid BoNT-A is reserved for patients with specific indications (TMJ disc displacement with muscle spasm or severe protrusive movement pain).
Clinical Response Timeline
Value of Injecting Multiple vs Single Muscles
Many early studies targeted only the masseter muscles and showed some pain improvement. However, if a patient's pain involves the temporalis or lateral pterygoid, those muscles may continue to cause symptoms if left untreated. Evidence suggests combined muscle injections can enhance results for widespread TMD myalgia:
- Trials that injected both masseter and temporalis muscles tended to report greater overall pain relief and improvement in jaw function compared to masseter-only approaches.
- Temporalis injections are particularly valuable for patients reporting temple headaches or pain on temporalis palpation. A systematic review found combined masseter+temporalis BoNT-A yielded a higher percentage of patients with ≥50% pain reduction.
- The lateral pterygoid, while not routinely injected, can reduce TMJ disc derangement symptoms and improve jaw opening in patients with internal derangements.
- In bruxism-related TMD, BoNT-A to both muscles significantly decreased nocturnal EMG activity. One study reported bruxism events per hour dropped from ~5 to ~1.7 after BoNT-A in both masseter and temporalis.
Clinically, a balanced approach is taken: if a patient's pain is clearly localised to one muscle group, injections can be limited to that group. If multiple muscles are involved, treating them together yields the best chance of complete relief.
Safety and Adverse Effects
In TMD trials, no serious adverse events have been reported from BoNT-A injections. The incidence of systemic side effects is very low given the localised nature of treatment. Nonetheless, there are important dose-dependent local effects:
Muscle Weakness and Atrophy
Up to one-third of patients notice chewing is less forceful. Effect is dose-dependent and usually mild. Patients adapt by eating softer foods initially. Strength returns over 2–3 months as the toxin wears off.
Reduced Bite Force
Maximum bite force and occlusal strength are reduced. Masseter bite force is lowest at ~3 months post-injection and then starts to recover. Patients may feel their bite is less strong for tough foods.
Speech or Smile Changes
If toxin diffuses to adjacent muscles (risorius or zygomaticus), patients can experience minor changes in facial expression (~15% in one series). Mild and resolve within weeks. Proper technique minimises this risk.
Injection Site Reactions
About 10–20% report short-lived injection site pain or mild localised swelling. Using ice and fine needles reduces this. Infection is exceedingly rare.
Bone Density/Joint Changes
Repeated high-dose BoNT-A may cause bone remodelling or resorption. One pilot study found decreased mandibular cortical bone density after multiple injections. Reinforces using the minimal effective dose.
Systematic reviews conclude that BoNT-A is safe and well-tolerated in the TMD population, with no significant increase in adverse events compared to placebo injections. Most side effects are localised and transient.
Conclusion
The off-label use of botulinum toxin type A has become an intriguing option for chronic orofacial pain, particularly TMD-related myofascial pain and bruxism-associated soreness. BoNT-A can provide meaningful pain relief and functional improvement in a subset of patients, especially in the first 1–3 months post-injection, and may outperform some traditional therapies in the short term. It is most likely to benefit those with muscle-dominant pain who have not responded adequately to occlusal splints, bite guards, anti-inflammatory medications, or physiotherapy.
However, BoNT-A is not a first-line treatment for most patients with orofacial pain. The overall research findings do not demonstrate a long-term advantage over conservative modalities for the average TMD patient. Some patients are "non-responders," and nearly all require repeat injections to maintain relief. Current clinical guidelines suggest it be used on a case-by-case basis when standard treatments fail or are contraindicated.
Clinical Decision Algorithm
References
- Saini RS, et al. PLoS One. 2024;19(3):e0300157. Systematic review/meta-analysis: BoNT-A vs placebo/others in TMD.
- Machado D, et al. J Pain. 2020;21(3-4):281-293. Systematic review/meta-analysis (12 RCTs): BoNT-A slight pain benefit at 1 month.
- Buzatu R, et al. Dent J (Basel). 2024;12(6):156. Systematic review (9 RCTs): BoNT-A in sleep bruxism-related TMD.
- Li K, et al. J Oral Rehabil. 2023;50(1). Meta-analysis: BoNT-A effective vs placebo up to 6 months.
- Ernberg M, et al. Pain. 2011;152(8):1988-1996. Multicenter RCT: BoNT-A (50 U) vs placebo in persistent myofascial TMD pain.
- Al-Wayli H. J Clin Exp Dent. 2016;8(5):e571-e577. RCT: BoNT-A vs conventional therapy for bruxism TMD pain.
- Guarda-Nardini L, et al. Cranio. 2012;30(2):95-102. RCT: BoNT-A vs fascial manipulation physiotherapy in myofascial pain.
- De la Torre Canales G, et al. Toxins. 2020;12(6):395. RCT (100 patients): BoNT-A vs saline vs splint over 6 months.
- Mor N, et al. Toxins. 2015;7(8):2791-2800. Review: BoNT-A in myofascial TMD – mechanism, injection technique, doses.
- Bouloux GF, et al. Front Oral Maxillofac Med. 2022;4:4. Review: Botox in oral/facial conditions.
Earn 1 AiCE Point – 100% Free
FREEComplete a 10-question assessment and short clinical reflection to earn your CPD certificate for this article.
